 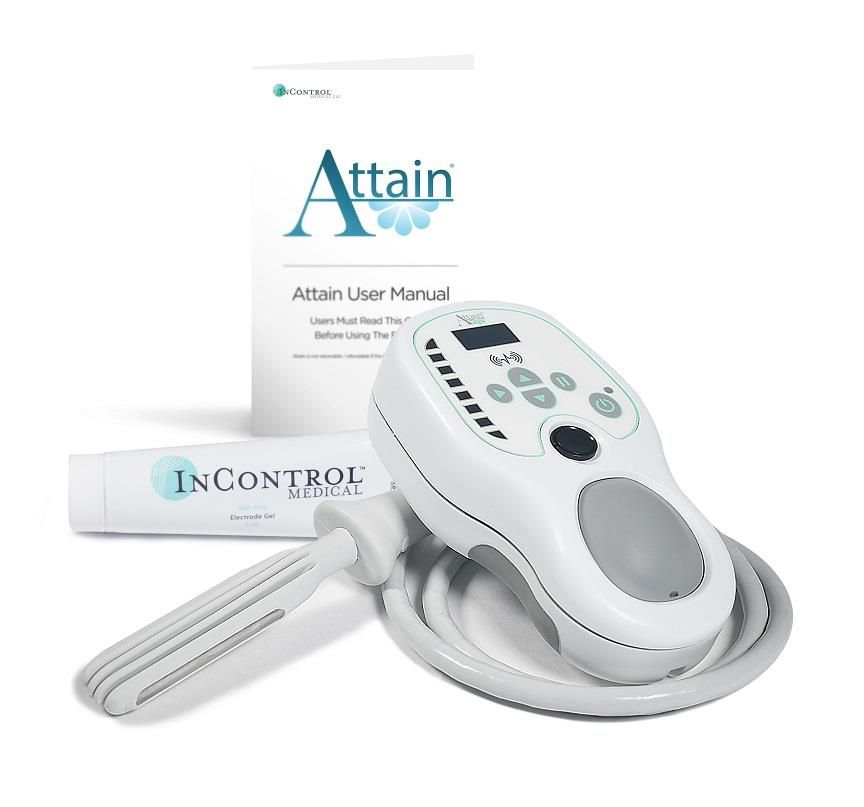
“Gynaecology societies recommend that women do pelvic floor muscle training as the first line against urinary incontinence, but the problem with Kegels is that you can’t visualise that part of your anatomy like you can when you’re doing a bicep curl. “I think the beauty of leva is that it makes first-line treatment accessible to women,” says Maus. While the final study has yet to be published, Maus says it has yielded “fabulous results.” This study only included 23 women, but Renovia has recently completed a 350-participant strong trial, investigating how well the device holds up compared to a standard pelvic floor exercise routine. By week six of the trial, 87% of women reported no urinary leakage at all. While it can take months to see progress from pelvic floor exercises, a clinical trial of the first-generation leva device saw 50% of women experience symptom improvement within a week. The change in incline can be seen on-screen in real-time by the user, allowing them to objectively assess their progress as they continually use the device. 
The vagina is lifted by the surrounding muscles during pelvic floor exercises, and leva measures the incline of that lift using its six sensors. The leva device assesses the Kegels being carried out by the user through six internal accelerometers. So, for two and a half minutes twice a day, you can get your pelvic floor health back.” How does the leva device help with incontinence? It coaches them to relax and rest and take a deep breath and to then contract their vagina and hold for fifteen seconds, which they can see visualised on the device. Renovia CEO Eileen Maus says: “What the patient does is insert the probe vaginally, and the connected app coaches them through the process of doing pelvic floor muscle therapy. This report also provides an in-depth analysis of the medical device market's opportunities and challenges, the influence of COVID-19 on the market, and government activities aimed to develop the APAC medical device market. 
The growth in medical device clinical trials in the APAC region is attributed to an increase in research partnerships between biopharma companies and clinical research organizations (CROs). The APAC medical device industry witnessed significant developments, launches, implementations, and adoption of new medical device technologies in the past decade, however the high cost of devices, regulatory hurdles, reimbursement pressure, and healthcare budget constraints have challenged market expansion across various territories and segments.ĪPAC is the fastest growing regional market for medical device clinical trials, with China and Japan emerging as major clinical trial hotspots. GlobalData's APAC Medical Devices Industry Landscape – 2022 free study breaks down the APAC medical devices market by segment and region and includes insights on local and global competitors, pipeline product developments, clinical trials studies, and significant deals in the industry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
